
The O―H distance ( bond length) is 95.7 picometres (9.57 × 10 −11 metres, or 3.77 × 10 −9 inches). The two hydrogen atoms are bound to the oxygen atom at an angle of 104.5°. The water molecule is not linear but bent in a special way. The root of these anomalies lies in the electronic structure of the water molecule. In its solid form, ice, water is less dense than when it is liquid, another unusual property.
For example, its melting point, 0 ☌ (32 ☏), and boiling point, 100 ☌ (212 ☏), are much higher than would be expected by comparison with analogous compounds, such as hydrogen sulfide and ammonia. Witness an experiment explaining why fresh water and seawater have different freezing points Contunico © ZDF Enterprises GmbH, Mainz See all videos for this articleĪlthough its formula (H 2O) seems simple, water exhibits very complex chemical and physical properties. For other major treatments of water, see climate environmental works hydrosphere ice and pollution. This article describes the molecular structure of water as well as its physical and chemical properties. Because seawater contains large quantities of dissolved salts, it must be desalinated for most uses, including human consumption. For example, the water used in high-pressure boilers must be at least 99.999998 percent pure.

Interestingly, the purity requirements of water for industrial use often exceed those for human consumption. As Earth’s population grows and the demand for fresh water increases, water purification and recycling become increasingly important. The water on the surface of Earth is found mainly in its oceans (97.25 percent) and polar ice caps and glaciers (2.05 percent), with the balance in freshwater lakes, rivers, and groundwater. ( A Britannica Publishing Partner) See all videos for this article Water harvesting Displayed by permission of The Regents of the University of California.
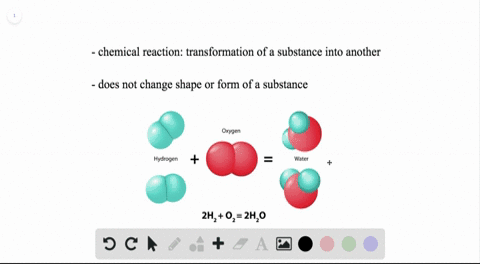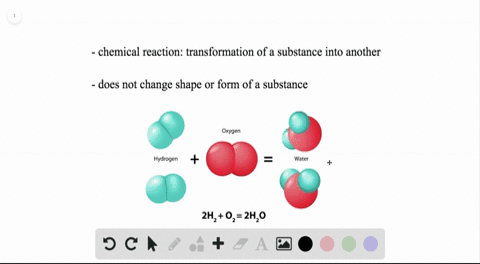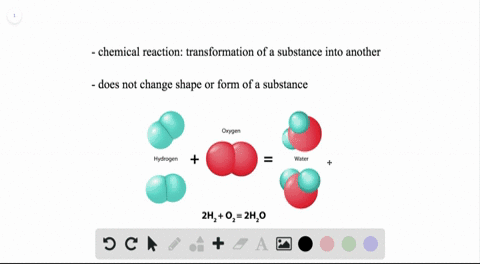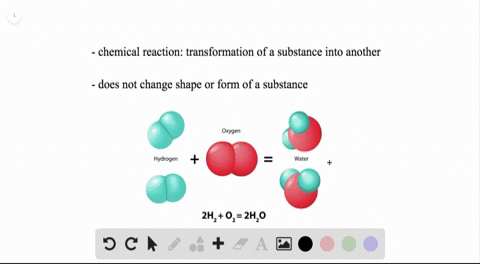
WHEN TWO WATER MOLECULES JOIN TOGETHER HOW TO
